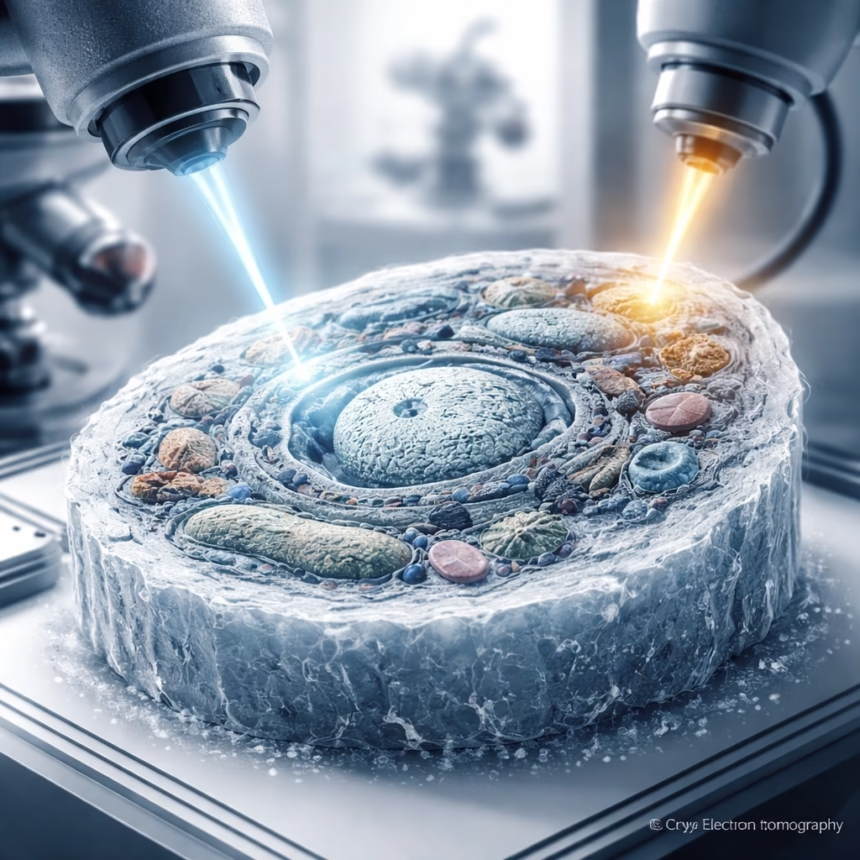
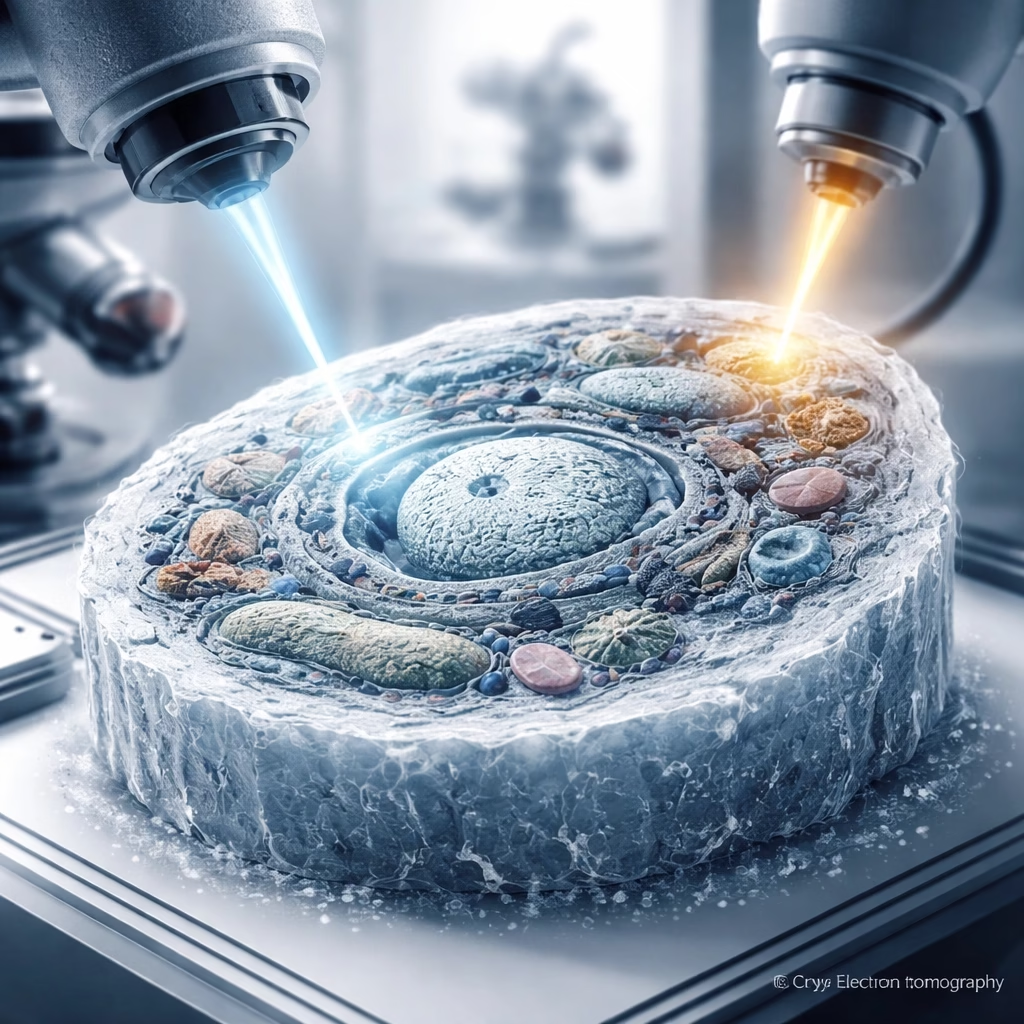
A collaborative team of researchers, primarily from the Institute for Structural Biology in Zurich, Switzerland, has unveiled a groundbreaking method for preparing biological samples, significantly enhancing the resolution and clarity of cryo-electron tomography (cryoET) imaging. Announced in late 2023, this innovative combined approach promises to revolutionize our understanding of cellular architecture and function by providing unprecedented views of subcellular components in their native state.
A Window into Life’s Core: The Cryo-ET Challenge
Cryo-electron tomography (cryoET) stands as a pivotal technique in structural biology, offering three-dimensional views of cellular components at near-atomic resolution. Unlike traditional electron microscopy, cryoET images samples plunged into liquid ethane, freezing them so rapidly that water molecules vitrify, preventing damaging ice crystal formation. This crucial step preserves biological structures in their native, hydrated state, providing a true snapshot of life within the cell.
Despite its immense power, cryoET faces a significant hurdle: sample thickness. Electron beams penetrate only very thin samples effectively. For whole cells or thicker cellular regions, the electron signal weakens and scatters excessively, leading to blurry images and the "missing wedge" problem, an artifact of limited tilt angles. This limitation has historically restricted high-resolution cryoET to smaller samples or the periphery of larger cells.
To overcome this, scientists developed methods to create ultrathin slices, known as lamellae, from frozen cells. The most prominent technique involves focused ion beam (FIB) milling, often integrated into a scanning electron microscope (SEM) system (cryo-FIB-SEM). Developed extensively in the early 2000s for materials science, cryo-FIB milling was adapted for biological samples, allowing a finely focused beam of gallium ions to precisely mill away material, sculpting a thin lamella, typically 100-300 nanometers thick, from a vitrified cell.
While cryo-FIB milling represented a monumental leap, it still presented challenges. The ion beam itself can induce damage to the delicate biological material, leading to amorphous layers on the lamella surface. Achieving perfectly uniform thickness across the entire lamella, especially for larger areas, remained difficult. Furthermore, precise targeting of specific organelles or regions within a dense cellular environment often required time-consuming and iterative milling steps, sometimes resulting in suboptimal lamellae or even sample loss.
The Breakthrough: A Synergistic Approach to Sample Preparation
The new method, developed by Dr. Elena Petrova and her team at the Institute for Structural Biology, addresses these long-standing limitations by integrating several advancements into a cohesive workflow. Dubbed the "Precision Cryo-Sculpting" technique, it combines enhanced cryo-immobilization, a multi-stage adaptive milling process, and a novel post-milling refinement step.
The first component involves an improved cryo-immobilization strategy. Utilizing high-pressure freezing coupled with targeted fluorescent markers, the researchers can precisely identify and tag specific organelles or regions of interest within a cell before the freezing process. This pre-localization significantly streamlines the subsequent milling steps, reducing the need for extensive search and navigation under the ion beam.
The core innovation lies in the multi-stage adaptive cryo-FIB milling process. Instead of a single, continuous milling operation, the Precision Cryo-Sculpting technique employs an initial rough milling phase using a higher ion current to quickly remove bulk material. This is followed by a series of progressively finer milling steps, utilizing significantly lower ion currents and adaptive beam control algorithms. These algorithms, developed in collaboration with instrumentation partners, monitor the lamella thickness and surface quality in real-time, adjusting the ion beam parameters to ensure exceptional uniformity and minimize damage. The team reports achieving lamellae with thickness variations of less than 5% across a 20×20 micron area.
Finally, a novel post-milling refinement step sets this method apart. Immediately after the final milling pass, the lamella undergoes a brief, ultra-low energy electron beam annealing process within the cryo-FIB-SEM chamber. This gentle treatment helps to repair minor surface damage induced by the ion beam and removes any residual amorphous layers, resulting in an exceptionally clean and pristine lamella surface. This step is critical for maximizing signal-to-noise ratios in subsequent cryoET imaging.
The key advantages of the Precision Cryo-Sculpting technique are quantifiable: it consistently produces lamellae that are up to 20% thinner (down to 70-80 nm in some cases) than previous methods, with a reported 50% reduction in surface damage artifacts. Furthermore, the improved targeting and adaptive milling reduce the overall preparation time for complex samples by approximately 30-40%, making high-quality lamella creation more efficient and reproducible.
Unlocking New Biological Frontiers: Impact and Applications
The implications of the Precision Cryo-Sculpting technique are profound for a wide array of biological disciplines. Structural biologists will now be able to visualize macromolecular complexes *in situ* with unprecedented clarity and resolution. This means obtaining clearer images of ribosomes synthesizing proteins, proteasomes degrading misfolded proteins, or molecular motors interacting with cytoskeletal filaments – all within their native cellular context, rather than as isolated components.
Cell biologists stand to gain new insights into dynamic cellular processes. The ability to prepare thinner, less damaged lamellae from specific regions will enable better visualization of membrane fusion events, viral budding from host cells, the intricate rearrangements of the cytoskeleton during cell division, and the complex interactions between organelles. Understanding these processes at a molecular level is crucial for deciphering fundamental cellular mechanisms.
Beyond basic research, the new method holds significant promise for understanding disease mechanisms and accelerating drug discovery. Researchers can now obtain sharper, more detailed views of pathogen-host interactions, such as how viruses hijack cellular machinery or how bacteria form biofilms. In cancer research, it could reveal subtle architectural changes in tumor cells or the precise locations of therapeutic targets. For neurodegenerative diseases, visualizing protein aggregates within neurons with higher fidelity could provide critical clues to disease progression.
Drug developers will particularly benefit from the ability to identify drug targets and understand drug action at a molecular level with greater precision. By visualizing drug-target interactions directly within the cellular environment, researchers can gain a more accurate picture of compound efficacy and specificity, potentially accelerating the development of new therapies. The interdisciplinary nature of this impact underscores its potential to bridge gaps between structural biology, cell biology, and biomedical research.
The Road Ahead: Future Milestones and Expansion
The immediate next steps for the Precision Cryo-Sculpting technique involve its wider adoption and the standardization of its protocol across leading cryo-EM facilities globally. Dr. Petrova's team is actively collaborating with other research institutions and instrument manufacturers to disseminate the methodology and integrate the adaptive milling algorithms into commercial cryo-FIB-SEM platforms.
Looking ahead, researchers anticipate further advancements, including the potential for increased automation of the entire lamella preparation workflow. This could enable higher throughput screening, making it feasible to analyze larger numbers of samples or screen for specific cellular states more efficiently. Efforts are also underway to explore the creation of even thinner lamellae, potentially pushing the resolution limits of cryoET further, or to develop methods for preparing larger lamellae that span multiple cells or even small tissue sections, offering a more holistic view of cellular communities.
The application of this technique to more complex biological systems, such as intact tissues or small multicellular organisms, represents a significant future milestone. This would allow for the study of cellular interactions within their physiological context, moving beyond isolated cells to understand systemic biology at a nanoscale. Collaborative efforts with advanced microscope manufacturers will continue to refine both the hardware and software, ensuring that the preparation techniques keep pace with improvements in electron detector and imaging technologies.
Ultimately, the long-term vision is to contribute to a comprehensive, high-resolution atlas of cellular life, mapping the intricate molecular landscape of various cell types, states, and disease conditions. The Precision Cryo-Sculpting technique is a crucial step towards realizing this ambitious goal, offering an unprecedented window into the fundamental processes that govern life.